Dr. Cecile Sebastian, a provider of Vivos appliances in Hawaii, shares her excitement about the recent advancement in sleep apnea treatment: “We are so excited to share this news! So many sleep apnea patients are undiagnosed and untreated. Dentists have had a pivotal role in screening patients to bring the numbers down. We are one of two Vivos appliance providers in Hawaii! Dr. Eugene Azuma and I are tirelessly working to help all sleep apnea sufferers from children to adults, especially those unable to tolerate CPAP machines. Please visit our sleep apnea page, or contact us for more information.”
Dr. Cecile Sebastian, a provider of Vivos appliances in Hawaii, shares her excitement about the recent advancement in sleep apnea treatment: “We are so excited to share this news! So many sleep apnea patients are undiagnosed and untreated. Dentists have had a pivotal role in screening patients to bring the numbers down. We are one of two Vivos appliance providers in Hawaii! Dr. Eugene Azuma and I are tirelessly working to help all sleep apnea sufferers from children to adults, especially those unable to tolerate CPAP machines. Please visit our sleep apnea page, or contact us for more information.”
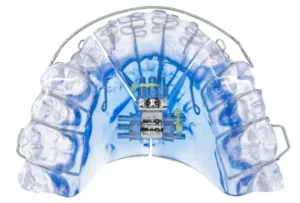
Following this enthusiasm from the medical community, Vivos Therapeutics, Inc., a leader in sleep-related breathing disorder treatments, has achieved a significant breakthrough. The company announced receiving the FDA’s 510(k) clearance for its CARE oral appliances, specifically designed to treat severe obstructive sleep apnea (OSA) in adults. This includes their DNA, mRNA, and mmRNA appliances, marking a major step forward in non-invasive OSA treatment options. Vivos now stands as the first to offer an alternative to CPAP machines and surgical neurostimulation implants for patients with severe OSA, building upon their earlier clearance for treating mild-to-moderate OSA.
Dr. David McCarty, a prominent medical sleep specialist, underscores the significance of this development. He emphasized the transformative potential this FDA approval holds for severe OSA patients who have found other treatments ineffective. McCarty highlighted the importance of understanding the oral vault’s role in OSA and praised Vivos’ approach to airway-centered dentistry.
Kirk Huntsman, Chairman and CEO of Vivos, elaborates on the wider implications of this FDA clearance. He envisions increased acceptance of Vivos treatments within the medical community, leading to more patient referrals and collaborations with medical professionals. Huntsman also anticipates enhanced relationships with third-party distributors and improved insurance reimbursement scenarios. This approval is seen as a critical milestone in expanding Vivos’ revenue growth prospects.
Clinical data submitted to the FDA showed encouraging results, with the majority of severe OSA patients experiencing significant improvements after using Vivos appliances. The treatment outcomes for severe OSA were notably more favorable than those for milder cases. In a separate study published in the Journal of Sleep Medicine, one in four Vivos patients experienced complete resolution of their OSA symptoms, demonstrating the effectiveness of Vivos treatments over a shorter period compared to lifelong interventions required by traditional methods like CPAP or surgical implants.